Max_B
Member
Thought this was a topical letter from 2014, considering the recent discussions on here between myself and Tim, which might be of interest...? I stumbled across it when I was researching something else.
I've also quoted from paper [3]... which shows stark differences in how thalamus neurons respond to oxygen and glucose deprivation, in comparison to deeper brain neurons within the hypothalamus.

[3] Ischemic vulnerability of the brain decreases rostrocaudally
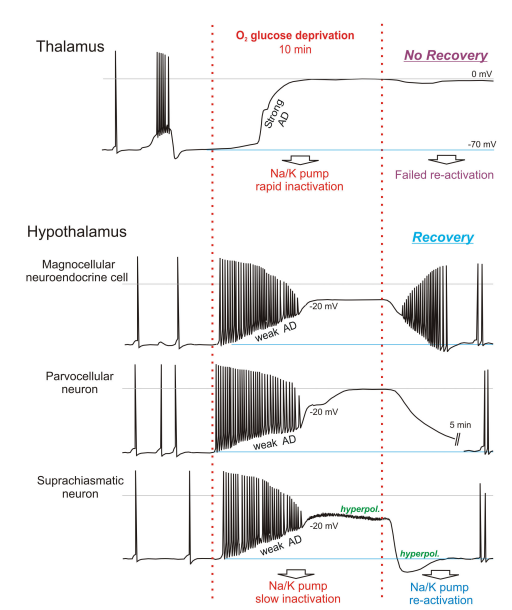
I've also quoted from paper [3]... which shows stark differences in how thalamus neurons respond to oxygen and glucose deprivation, in comparison to deeper brain neurons within the hypothalamus.

[3] Ischemic vulnerability of the brain decreases rostrocaudally
Magnetic Resonance imaging of patients who survive global ischemia but remain in a persistent vegetative state (PVS), show relatively normal brainstem function but dysfunctional higher brain activity [5] [6] [30] [7], a finding supported by numerous studies measuring regional metabolism [8]. There is also evidence from animal studies that anoxic depolorisation (AD) which is acutely damaging in higher brain [31] is comparatively weak in brainstem. Specifically, AD arising from respiratory arrest in intact rat brain measured using K + -sensitive electrodes [17] showed that the profile of elevated [K + ] o (representing AD strength) is delayed, is slower to rise, and peaks at lower levels in hypothalamus and brainstem compared to cortex and striatum. As well, global ischemia in dogs for 20 minutes [11] evoked a gradient of neuronal injury assessed 7 days later as follows: neocortex, hippocampus, cerebellum > basal ganglia> thalamus > brainstem. In fact, they detected no injury in midbrain, pons or medulla despite all regions being deprived of blood over the same period. The adult hypothalamus and brainstem do not naturally undergo spreading depression [12] so it is not surprising that they are also less supportive of AD.
Neurons in higher brain undergo strong AD (see Introduction) while hypothalamic and brainstem neurons show weak or no AD [15] [16][32]. This also holds for the less metabolically stressed version of AD, spreading depression (ibid) and in slices is obviously independent of regional variation in blood flow. Specifically, using whole-cell patch and 2-photon microscopy, we have previously reported a clear difference in neuronal responses to Oxygen/glucose deprivation between susceptible neocortical pyramidal neurons and more resilient Magnocellular Neuroendocrine Cells in the hypothalamic supraoptic nucleus [15]. Here we examined if and where a demarcation in susceptibility to anoxic depolorisation and spreading depression generation can be identified. We show that the transition from vulnerable to more resilient is at the thalamus-hypothalamus interface. Following 10 minutes of Oxygen/glucose deprivation in rodent slices, the thalamus is essentially dead. No new thalamic neurons can be obtained post-Oxygen/glucose deprivation, even following up to 1 hour recovery in control artificial cerebral spinal fluid. Thalamic neurons, like those of neocortex [33], CA1 hippocampus [34] [35] [36] and striatum [2] undergo strong and irreversible AD that kills the neurons in brain slices.
In contrast, tens of microns away in hypothalamus we recorded three neuronal types in the paraventricular nucleus (PVN) and suprachiasmatic nucleus (SCN) showing comparative resilience to 10 or 15 minutes of Oxygen/glucose deprivation. These neurons recover their membrane potential, input resistance and action potential amplitude. Moreover, newly acquired neurons are readily obtained in the hypothalamus following Oxygen/glucose deprivation, a feat not possible in adjacent thalamus, even following up to an hour of recovery in control artificial cerebral spinal fluid. Their resting membrane potential, input resistance and action potential amplitude are similar to their neuron counterparts not previously exposed to Oxygen/glucose deprivation, further indicating good recovery from Oxygen/glucose deprivation. This resilience is similar to that reported in a fourth hypothalamic region, the supraoptic nucleus [15]. That study sampled only one type of `higher` (neocortical) neuron and one type of `lower` hypothalamic Magnocellular Neuroendocrine neuron but there was no way to generalize that hypothalamic neurons resist ischemic resistance. By sampling 3 additional hypothalamic neuronal types and comparing them to nearby thalamic neurons, this study can make that conclusion.
Neurons in higher brain undergo strong AD (see Introduction) while hypothalamic and brainstem neurons show weak or no AD [15] [16][32]. This also holds for the less metabolically stressed version of AD, spreading depression (ibid) and in slices is obviously independent of regional variation in blood flow. Specifically, using whole-cell patch and 2-photon microscopy, we have previously reported a clear difference in neuronal responses to Oxygen/glucose deprivation between susceptible neocortical pyramidal neurons and more resilient Magnocellular Neuroendocrine Cells in the hypothalamic supraoptic nucleus [15]. Here we examined if and where a demarcation in susceptibility to anoxic depolorisation and spreading depression generation can be identified. We show that the transition from vulnerable to more resilient is at the thalamus-hypothalamus interface. Following 10 minutes of Oxygen/glucose deprivation in rodent slices, the thalamus is essentially dead. No new thalamic neurons can be obtained post-Oxygen/glucose deprivation, even following up to 1 hour recovery in control artificial cerebral spinal fluid. Thalamic neurons, like those of neocortex [33], CA1 hippocampus [34] [35] [36] and striatum [2] undergo strong and irreversible AD that kills the neurons in brain slices.
In contrast, tens of microns away in hypothalamus we recorded three neuronal types in the paraventricular nucleus (PVN) and suprachiasmatic nucleus (SCN) showing comparative resilience to 10 or 15 minutes of Oxygen/glucose deprivation. These neurons recover their membrane potential, input resistance and action potential amplitude. Moreover, newly acquired neurons are readily obtained in the hypothalamus following Oxygen/glucose deprivation, a feat not possible in adjacent thalamus, even following up to an hour of recovery in control artificial cerebral spinal fluid. Their resting membrane potential, input resistance and action potential amplitude are similar to their neuron counterparts not previously exposed to Oxygen/glucose deprivation, further indicating good recovery from Oxygen/glucose deprivation. This resilience is similar to that reported in a fourth hypothalamic region, the supraoptic nucleus [15]. That study sampled only one type of `higher` (neocortical) neuron and one type of `lower` hypothalamic Magnocellular Neuroendocrine neuron but there was no way to generalize that hypothalamic neurons resist ischemic resistance. By sampling 3 additional hypothalamic neuronal types and comparing them to nearby thalamic neurons, this study can make that conclusion.


